
The factor-label method yields the desired cancellation of units, and the computed result is on the order of 10 22 as expected. The process should be very similar with other such atoms, just make sure to keep your periodic table and calculator handy.\] Then, I used "Avogadro's Number", or #6.022 * 10^23 " atoms per mole"# to convert the mole amount to atoms of Carbon. #4.01 " g Carbon" ((1 " mol Carbon")/(12.01 " g Carbon")) ((6.022 * 10^23 atms Carbon)/(1 " mol Carbon")) = 2.01 * 10^23 " atms Carbon"#īasically, I first wrote down the amount in grams, and I used the molar mass of Carbon (which can be found on the periodic table under Carbon) 12.01 g/mol to convert 4.01 g of Carbon to moles of Carbon. Next, using the periodic table, find the atomic mass in g/mol of each element (the molar mass of an element is equal to its atomic mass): Molar Mass (g/mol) H (Hydrogen) 1.00794: S (Sulphur/Sulfur) 32.065: O (Oxygen) 15.9994: 3. 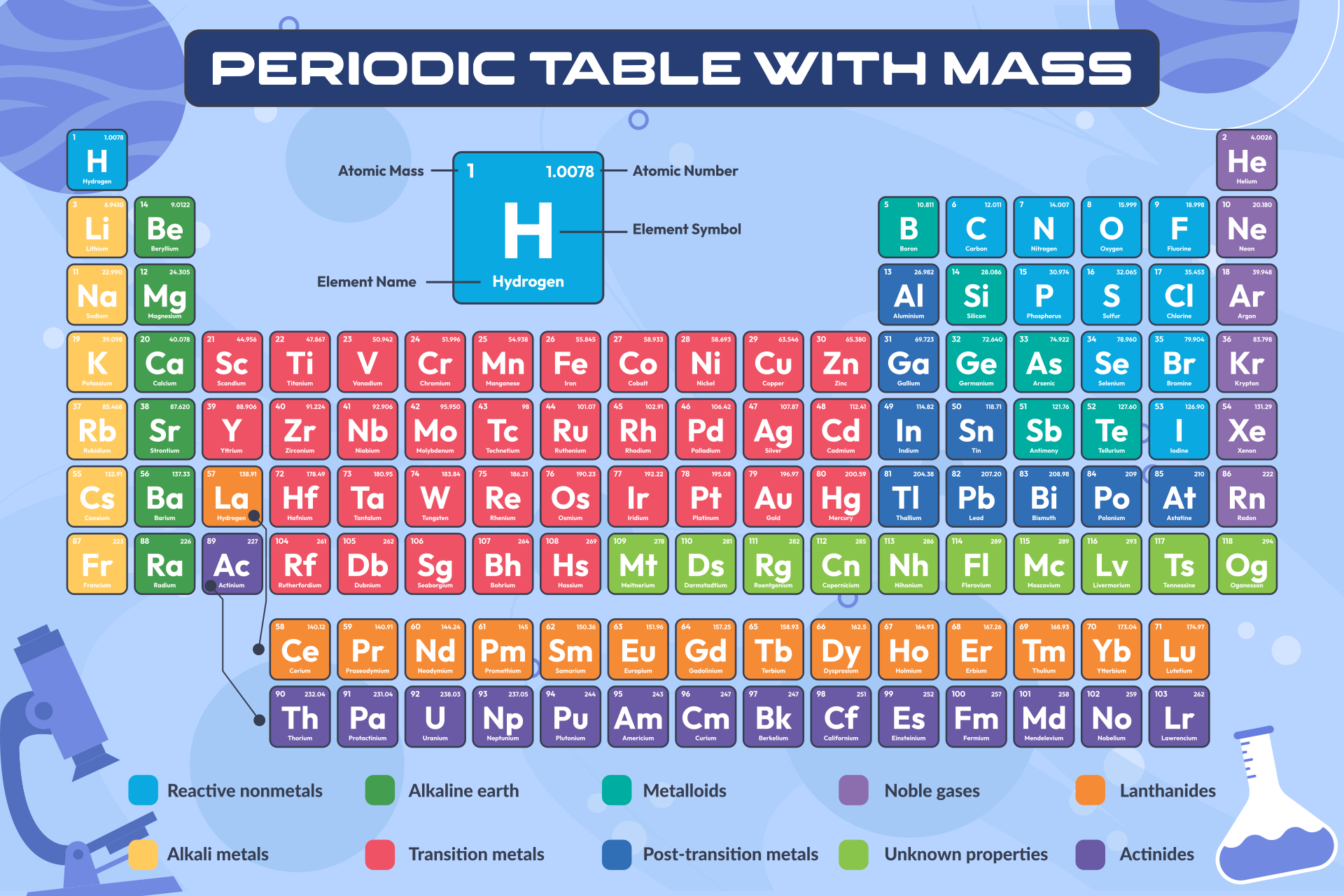 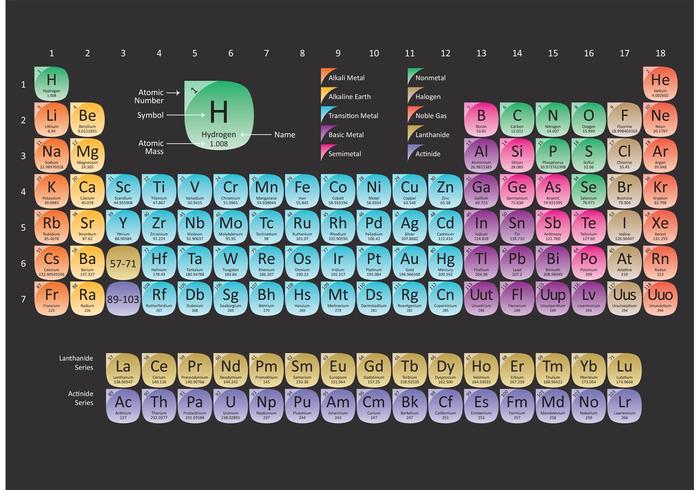
The breakdown would be as follows, with dimensional anaysis: It can supply you with an accurate representation of a substance’ssize and mass, and valence electron shell. And, lets assume that you are given 4.01 g of Carbon, and you are tasked to find the number of atoms in that mass of Carbon. Periodic Table Of Elements Rounded Atomic Mass The Regular Table is a crucial part of the research into scientific research, also it can be useful in discovering a substance’s qualities. For this case, lets say that that substance is Carbon (C). Prepare a concept map and use the proper conversion factor. Let's say that you are given an amount of grams of a substance. Number of atoms are calculated using a number known as "Avogadro's number." How many atoms of zinc, Zn, are in #"120.71 g Zn"?# Once you have moles, multiply by Avogadro's number to calculate the number of atoms. So, if you are given the mass of an element, you use the periodic table to find its molar mass, and multiply the given mass by the reciprocal of the molar mass. To calculate atoms of Au, multiply moles Au by Avogadro's number. The final answer will be rounded to four significant figures. I'm keeping a couple of guard digits to reduce rounding errors. Weve discussed mass at length by doing molar mass conversions. And we can do this for all the elements in the periodic table. For potassium then, it is 39.098 g/mol which means one mole of potassium atoms weighs 39.098 g. Transcribed image text: Calculate the molar mass of each element or compound below. #58.27color(red)cancel(color(black)("g Au"))xx(1"mol Au")/(196.967color(red)cancel(color(black)("g Au")))="0.295836 mol Au"# What’s important for today’s topic is that numerically, the molar mass is equal to the average atomic mass of the given atom, so we can look it up in the periodic table. To calculate moles of Au, multiply the given mass by the reciprocal of the molar mass. Use this visual tool for calculating molar mass for any chemical formula. The molar mass of copper (II) chloride dihydrate is g/mol. The molar mass of magnesium phosphate is g/mol. The molar mass of glucose C6H1206 is g/mol. This means that its molar mass is #"196.967 g/mol"#. Use a periodic table to fill in the following blanks: (please round off your answer to an interger and do not include a demical. The periodic table shows us that gold, Au, has the atomic weight 
How many atoms of gold are in #"58.27 g"# of gold? It is obtained chiefly from the mineral cassiterite, which contains tin dioxide. ~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~ Tin is a post-transition metal in group 14 of the periodic table. Every chemical element has a specific atomic number, which provides insight into the number of protons present within its nucleus. The following example will show you how to do that. The periodic table lists the atomic mass of carbon as 12. #"Mass"#rarr#"Moles"# and #"Moles"#rarr#"Atoms"# The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole. There are two basic steps to get from the given mass to the number of atoms. For calculations, use the given molar mass on the periodic table without rounding. Usually you will have a given mass of an element. Re: Rounding molar mass on the periodic table. The elements in group 1 are known as the alkali metals, those in group 2. Groups are numbered 118 from left to right. Elements can be classified as metals, metalloids, and nonmetals, or as a main-group elements, transition metals, and inner transition metals. One mole of anything, including atoms, is #6.022xx10^23# (Avogadro's number) of them. Elements in the same group of the periodic table have similar chemical properties.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
